Understanding how to reconstitute peptides correctly is one of the most important skills in research peptide handling. Improper reconstitution degrades peptide integrity, produces inaccurate concentrations, and compromises research results. This complete guide covers everything researchers need to know, from solvent selection through storage of reconstituted peptides, based on established laboratory best practices.
What Is Peptide Reconstitution
Lyophilized peptides are supplied as freeze-dried powder to maximise stability during storage and shipping. Before use in research applications the powder must be dissolved in an appropriate solvent to create a liquid solution at a known concentration. This process is called reconstitution.
The reconstitution process requires sterile technique, appropriate solvent selection, and precise volume calculation to produce a solution of the intended concentration. Errors at any of these steps affect downstream research accuracy.
What You Need Before Starting
Gather all materials before beginning the reconstitution process. Working with incomplete equipment increases contamination risk and handling errors.
Required materials:
Sterile bacteriostatic water or sterile saline solution Alcohol wipes or isopropyl alcohol at 70% concentration Sterile syringes — 1ml insulin syringes for precise measurement Sterile needles — 23 to 27 gauge for drawing and injecting solvent The peptide vial at room temperature A clean sterile working surface Research documentation for recording concentration and date
Optional but recommended:
Sterile gloves Magnifying glass for reading vial markings accurately Calculator for concentration verification
Choosing the Right Solvent
Solvent selection is the first critical decision in reconstitution. The wrong solvent can degrade the peptide, affect solubility, or compromise research validity.
Bacteriostatic Water — The Standard Choice
Bacteriostatic water contains 0.9% benzyl alcohol which inhibits bacterial growth and extends the usable life of reconstituted peptides at refrigerator temperatures. It is the most commonly used solvent for research peptide reconstitution and is appropriate for the majority of compounds.
Use bacteriostatic water when:
- The peptide will be stored for more than a few days after reconstitution
- Extended shelf life of the reconstituted solution is required
- The research protocol permits benzyl alcohol in the solvent
Sterile Water for Injection
Sterile water contains no preservatives and is appropriate for immediate use applications where the entire reconstituted volume will be used promptly. Without benzyl alcohol, bacterial growth is not inhibited and the reconstituted solution degrades faster.
Use sterile water when:
- The entire solution will be used within 24 hours
- The research protocol specifically excludes benzyl alcohol
- Sensitivity to preservatives is a concern in the study design
Sterile Saline — 0.9% Sodium Chloride
Sterile saline is an isotonic solution sometimes preferred for specific peptide types or research protocols. It provides no antimicrobial protection but is appropriate for certain applications.
Acetic Acid Solution
Some peptides including certain growth hormone fragments have poor solubility in aqueous solutions and require dilute acetic acid (typically 0.1% to 1%) for complete dissolution. If your peptide does not dissolve in bacteriostatic water acetic acid solution may be required. Check compound-specific documentation before proceeding.
| Peptide Type | Recommended Solvent |
|---|---|
| Most peptides | Bacteriostatic water |
| Immediate use | Sterile water |
| Poor aqueous solubility | 0.1% acetic acid |
| Specific protocols | Sterile saline |
Concentration Calculation — Before You Add Solvent
Calculating your target concentration before adding solvent is essential. Adding the wrong volume of solvent produces a solution at the wrong concentration, invalidating all downstream measurements.
The basic formula:
Concentration (mg/ml) = Peptide amount (mg) divided by Solvent volume (ml)
Practical example:
You have a 10mg vial of Tesamorelin and want a concentration of 2mg/ml.
Volume needed = Peptide amount divided by Target concentration Volume needed = 10mg divided by 2mg/ml Volume needed = 5ml of bacteriostatic water
Adding 5ml of bacteriostatic water to a 10mg vial produces a 2mg/ml solution.
Common concentration reference:
| Vial Size | Solvent Added | Resulting Concentration |
|---|---|---|
| 2mg | 1ml | 2mg/ml |
| 2mg | 2ml | 1mg/ml |
| 5mg | 1ml | 5mg/ml |
| 5mg | 2.5ml | 2mg/ml |
| 10mg | 2ml | 5mg/ml |
| 10mg | 5ml | 2mg/ml |
| 10mg | 10ml | 1mg/ml |
| 20mg | 4ml | 5mg/ml |
| 20mg | 10ml | 2mg/ml |
Calculate your specific concentration before proceeding and record it in your research documentation.
Step-by-Step Reconstitution Protocol
Follow these steps precisely. Do not skip steps or change the order.
Step 1 — Allow the vial to reach room temperature
Remove the peptide vial from the freezer. Allow it to sit at room temperature for 15 to 20 minutes before opening. Reconstituting a cold vial causes temperature differential issues and increases condensation risk inside the vial. Never open a cold vial directly from the freezer.
Step 2 — Prepare your working surface
Wipe your working surface with an alcohol wipe. Allow it to dry completely before placing any materials on it. A clean surface reduces airborne contamination risk during the reconstitution process.
Step 3 — Clean the vial tops
Wipe the rubber septum of both the peptide vial and the bacteriostatic water vial with a fresh alcohol wipe. Allow the alcohol to evaporate for 10 seconds before inserting any needle. This step eliminates surface contaminants that could enter the solution during needle insertion.
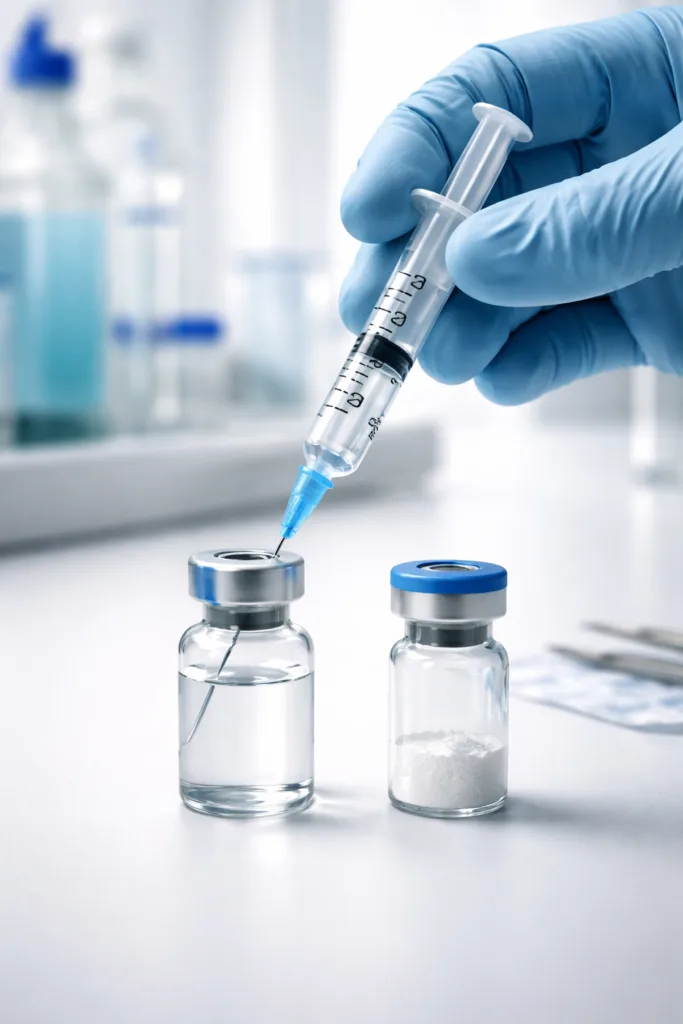
Step 4 — Draw the calculated solvent volume
Using a sterile syringe and needle, draw the calculated volume of bacteriostatic water from the water vial. If you calculated 5ml as your required volume draw exactly 5ml. Verify the volume by holding the syringe at eye level and reading the graduated markings accurately.
Step 5 — Add solvent slowly to the peptide vial
Insert the needle through the rubber septum of the peptide vial at a slight angle. Direct the solvent stream slowly down the inner glass wall of the vial. Do not aim the stream directly at the peptide powder. Adding solvent too quickly or directly onto the powder causes foaming, peptide degradation, and incomplete dissolution.
Add the solvent in small increments if working with larger volumes. Pause between additions to allow the solution to settle.
Step 6 — Gently swirl until dissolved
Remove the needle and gently swirl the vial in slow circular motions. Do not shake the vial. Do not vortex. Shaking and vortexing create mechanical stress that breaks peptide bonds and degrades the compound. Continue swirling gently until the powder is completely dissolved and the solution is clear.
Some peptides take longer to dissolve than others. If the solution is not clear after 2 to 3 minutes of gentle swirling, allow it to sit for an additional 5 minutes at room temperature and swirl again. Do not force dissolution by shaking.
Step 7 — Inspect the solution
Hold the reconstituted vial against a light source. The solution should be clear and free of particulates. Slight colour is acceptable for some peptides. Cloudiness, visible particles, or unusual colour may indicate incomplete dissolution or contamination. If the solution does not clear after extended gentle swirling contact sales@planetpeptide.co for guidance on that specific compound.
Step 8 — Label and document
Label the vial immediately with the reconstitution date, calculated concentration, and your initials. Record this information in your research documentation. This step is critical for research traceability and prevents concentration errors in downstream use.
Post-Reconstitution Storage
Storage conditions after reconstitution directly affect peptide stability and research validity.
Standard refrigerated storage:
Store reconstituted peptides at 2°C to 8°C — standard refrigerator temperature. This temperature range slows bacterial growth when using bacteriostatic water and minimises peptide degradation. Most reconstituted peptides remain stable for 4 weeks under these conditions.
Avoid the following:
Do not freeze reconstituted peptides repeatedly. Each freeze-thaw cycle introduces mechanical stress that degrades peptide structure. If you need to store reconstituted peptide for longer than 4 weeks divide the reconstituted solution into single-use aliquots before freezing and thaw only what is needed for each research session.
Keep reconstituted vials away from light. UV exposure degrades many peptide compounds. Store in a light-protected container or wrap vials in aluminium foil.
Storage duration reference:
| Storage Condition | Maximum Duration |
|---|---|
| 2 to 8°C with bacteriostatic water | 4 weeks |
| 2 to 8°C with sterile water | 24 to 72 hours |
| -20°C single-use aliquots | 3 to 6 months |
| -80°C single-use aliquots | Up to 12 months |
Common Reconstitution Mistakes and How to Avoid Them
Mistake 1 — Adding solvent directly to the powder
Adding bacteriostatic water directly onto the lyophilized powder causes foaming and can denature surface peptide molecules. Always direct the stream down the vial wall.
Mistake 2 — Shaking the vial
Mechanical shaking breaks peptide bonds through agitation. Always swirl gently and never vortex or shake.
Mistake 3 — Not allowing the vial to warm to room temperature
Cold vials create condensation issues and thermal stress during reconstitution. Always wait 15 to 20 minutes after removing from the freezer.
Mistake 4 — Incorrect concentration calculation
Calculate concentration before adding solvent. Write it down. Verify it independently. Concentration errors invalidate research data and waste expensive research compounds.
Mistake 5 — Not labelling the vial immediately
Unlabelled vials create serious research documentation problems. Label every vial at the moment of reconstitution without exception.
Mistake 6 — Using the wrong solvent
Check solubility requirements for each specific peptide before reconstituting. Most dissolve in bacteriostatic water but some require acetic acid. Using the wrong solvent results in incomplete dissolution or peptide degradation.
Compound-Specific Reconstitution Notes
While the general protocol above applies to most peptides certain compounds have specific considerations researchers should be aware of.
BPC-157 — Dissolves readily in bacteriostatic water. Stable for 4 weeks refrigerated. Compatible with saline for specific applications.
TB-500 — May require gentle warming of the vial in your hand during swirling for complete dissolution. Dissolves fully in bacteriostatic water.
CJC-1295 — Dissolves in bacteriostatic water. The DAC component increases molecular weight and may require slightly longer swirling time for complete dissolution.
Tesamorelin — Dissolves in bacteriostatic water. Stable for 4 weeks refrigerated. The full 44-amino acid structure dissolves more slowly than shorter peptides — allow adequate time during step 6.
GHK-Cu — The copper complex dissolves readily and produces a characteristic blue-green colour in solution. This colour is normal and not a sign of contamination or degradation.
Semaglutide — Dissolves in bacteriostatic water. Due to molecular size allow additional swirling time. Protect from light during and after reconstitution.
For compound-specific guidance on any Planet Peptide research compound contact our scientific support team at sales@planetpeptide.co.
Research Safety and Compliance
All peptides supplied by Planet Peptide are intended strictly for laboratory research purposes. Handle reconstituted peptide solutions with appropriate personal protective equipment including gloves and eye protection. Follow your institution’s chemical handling and disposal protocols for all research materials.
Reconstituted solutions should be disposed of according to applicable laboratory waste disposal regulations. Do not dispose of research chemicals in general waste or down drains without following institutional guidelines.
Frequently Asked Questions
What is the best solvent for reconstituting peptides?
Bacteriostatic water is the recommended solvent for most research peptides. It contains 0.9% benzyl alcohol which inhibits bacterial growth and extends the usable life of reconstituted solutions to approximately 4 weeks when stored at 2 to 8°C.
How much bacteriostatic water should I add to a 10mg peptide vial?
The volume depends on your target concentration. For a 2mg/ml solution add 5ml to a 10mg vial. For a 5mg/ml solution add 2ml. Calculate your target concentration before adding any solvent using the formula: volume equals peptide amount divided by target concentration.
Why should I not shake the peptide vial during reconstitution?
Shaking creates mechanical agitation that breaks peptide bonds through a process called shear stress. This degrades the peptide and reduces research validity. Always swirl gently rather than shaking.
How long does reconstituted peptide last in the refrigerator?
Reconstituted peptides stored in bacteriostatic water at 2 to 8°C typically remain stable for 4 weeks. Peptides reconstituted in plain sterile water without a preservative should be used within 24 to 72 hours.
Can I freeze reconstituted peptides for longer storage?
Yes but only as single-use aliquots. Divide the reconstituted solution into individual use portions before freezing. Each aliquot should be used once after thawing and not refrozen. Repeated freeze-thaw cycles degrade peptide integrity.
What should I do if the peptide does not dissolve completely?
Allow the vial to sit at room temperature for an additional 5 to 10 minutes and swirl again gently. If the solution remains cloudy the compound may require a different solvent such as dilute acetic acid. Contact sales@planetpeptide.co for compound-specific guidance.
Does the colour of the reconstituted solution matter?
Most peptides produce a clear colourless solution. Some exceptions exist — GHK-Cu produces a characteristic blue-green colour which is normal. If your solution is cloudy, has visible particles, or has an unexpected colour that is not documented for that compound, it may indicate contamination or incomplete dissolution.